
Crosstalk signaling mechanisms that regulate intra-islet endothelial cells survival and peri-islet vessel formation: Role of novel intra-pancreatic microvessel fragments
Ahad A. Kodipad1, Krishna K. Samaga1, Chandrashekar B. Revanna1, Siddharth Narayanan1, Jaimie D. Nathan1, Appakalai N. Balamurugan1.
1Pediatrics and Surgery, Center for Clinical and Translational Research, Nationwide Children's Hospital / Ohio State University, Columbus, OH, United States
Introduction: Islet grafts have significantly less new vascular supply and low oxygen tension than normal islets because the isolation procedure strips the dense microvascular network of islets leading to poor engraftment after transplantation. This poor engraftment results in limited inosculation with the host circulation causing many islets to die due to poor revascularization. To overcome this problem, we introduce a novel method to use intra-pancreatic microvessel fragments (IPMVF). MVFs contain antigen (Sca)-1/VEGFR-2-positive endothelial progenitor cells and mesenchymal stem cells expressing common markers such as CD73, CD44, CD90 and CD117 and large quantities of endothelial cells (EC). Freshly isolated human islets in the standard culture media cannot form cellular sprouts. Intra-islet ECs undergo quiescent phase after human islet isolation. However, co-culture and co-transplantation studies demonstrate that IP-MVFs stimulate intra-islet EC sprouting to form peri-islet vessels (PIVs). In this study, we tested the role of TAK1 (transforming growth factor β-activated kinase), a mitogen activating protein kinase in islet EC survival.
Methods: Intra-pancreatic MVF’s and human islets were isolated from human pancreases (n=21). Freshly collected human islets and IP-MVFs (n=10; 1:1000), cultured in the vascular cell basal medium with endothelial cell growth supplements (ECGS). To understand the role of TAK1 in intra-islet sprouting, we used TAK1 inhibitor. The intra-islet EC sprouting phenotypes were assessed on pure human islets embedded in 3D-collagen gels in the presence of vehicle (DMSO) alone or TAK1 inhibitor (0.5µM-2μM; 5Z-7-oxozeaenol) .To assess the tip-cell specific targets during PIV formation in the control group (islets alone) versus the experimental group (islets + TAK1 inhibitor), we used a laser dissection microscope to isolate and confirm the presence of tip cells. Specifically, intra-islet ECs were transduced with control or TAK1 shRNA for 36h. The cells were washed and treated with recombinant vascular endothelial growth factor-A (VEGF-A) or fibroblast growth factor (FGF) at different time points and the cell lysates were analyzed for various proteins (n=5) by western blotting.
Results: Our results indicate that human islets in the presence of IP-MVF ECs, form peri-islet vessels (PIVs). This observation suggests that IP-MVF ECs release pro-angiogenic factors which stimulated intra-islet ECs to form PIVs. Various stimuli such as TGF-β and inflammatory cytokines (IL-1, TNF) activate TAK1. Multiple signaling cascade regulate intra-islet EC survival and sprout formation. Treatment with TAK1 inhibitor (5Z-7-oxozeaenol or RIN) showed a reduced level of sprouting in freshly isolated islets.
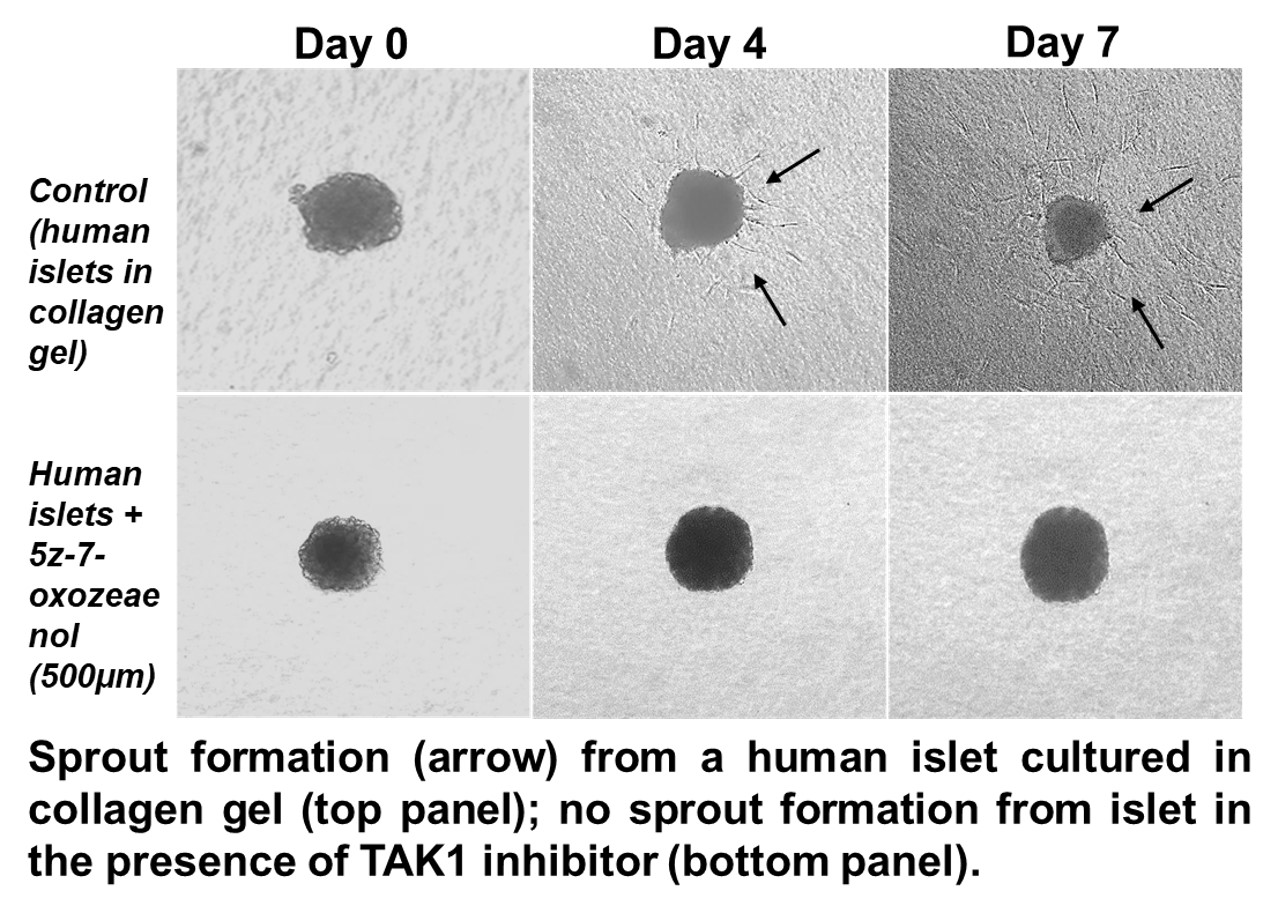
We confirmed that TAK1 regulated PIV sprouting in human islets through activating MAPK and NF-ƙB signaling pathways.
Conclusion: TAK1 regulates human intra-islet EC survival and PIV sprout formation through activating MAPK and NF-ƙB signaling associated with EC survival, migration, and proliferation, and that TAK1-mediated signaling pathways crosstalk with Notch signaling to promote human intra-islet EC sprouting. Our study results also determined how the Notch and TAK1 molecular crosstalk events stimulated intra-islet PIV sprouting to eventually aided the IP-MVF and islet-to-host inosculation and proved the efficacy of IP-MVFs as novel neovascularization units.